Added content from our newsletter of CRN updates exclusively for C-suite executives.
Contact Gretchen Powers (gpowers@crnusa.org) for more information about the newsletter.
CRN Promotes Responsible Innovation at FDA Public Meeting

At a pivotal moment for the future of dietary supplement innovation, two voices from CRN helped bring clarity—and urgency—to the conversation at FDA's March 27 public meeting on the evolving scope of dietary ingredients (Docket FDA-2026-N-2047).
CRN SVP and Chief Science Officer Andrea Wong, Ph.D., and SVP and General Counsel Megan Olsen delivered testimony that, together, bridged science, law, and practical reality in a way that reflects the unique value CRN provides.
Dr. Wong grounded the discussion in what innovation actually looks like today. From precision fermentation to next-generation probiotics, the science has moved far beyond 1994—but regulatory interpretation hasn’t always kept pace. Her message was clear: we cannot define “dietary substance” so narrowly that it excludes the very innovations DSHEA was designed to accommodate.
As she put it, responsible industry isn’t looking for shortcuts—it’s “knocking at the front door,” ready to demonstrate safety through the appropriate pathways, not forced into inefficient or impractical workarounds.
Ms. Olsen then reinforced that position with legal precision. The statute’s language—“to supplement the diet by increasing the total dietary intake”—was intentionally expansive. It does not require prior presence in the food supply, nor does it limit innovation to what existed decades ago. Legislative history confirms this was a conscious choice by Congress to allow the category to evolve alongside science. The path forward, as she emphasized, is not to rewrite the law—but to interpret it as written, in a way that supports both innovation and safety.
Together, their testimony reflects what CRN has consistently stood for:
✔️ Science-based policy
✔️ Regulatory clarity
✔️ Responsible innovation
✔️ A viable pathway (NDI) that actually works
As Dr. Wong noted, the detailed questions FDA is asking about identity, manufacturing, and composition are valid—but they belong in the evaluation of safety, not as a gatekeeping mechanism for whether an ingredient qualifies in the first place. This distinction matters, because when the framework is too narrow, innovation doesn’t stop—it just moves elsewhere, often outside the very oversight system designed to protect consumers. A more inclusive, science-aligned interpretation does the opposite: it brings innovation into the regulatory system, where it can be properly evaluated.
CRN, with its Regulatory Affairs Committee, submitted comments addressing the definition of “dietary substance,” access to the NDI pathway, emerging ingredient categories, and more. The outcome will shape both regulatory policy and the trajectory of innovation across proteins, enzymes, microbials—and beyond.
NEW: CRN Files Citizen Petition Urging FDA to Clarify DSHEA Disclaimer Placement for Dietary Supplements
Age Restriction Laws Could Spread State-by-State
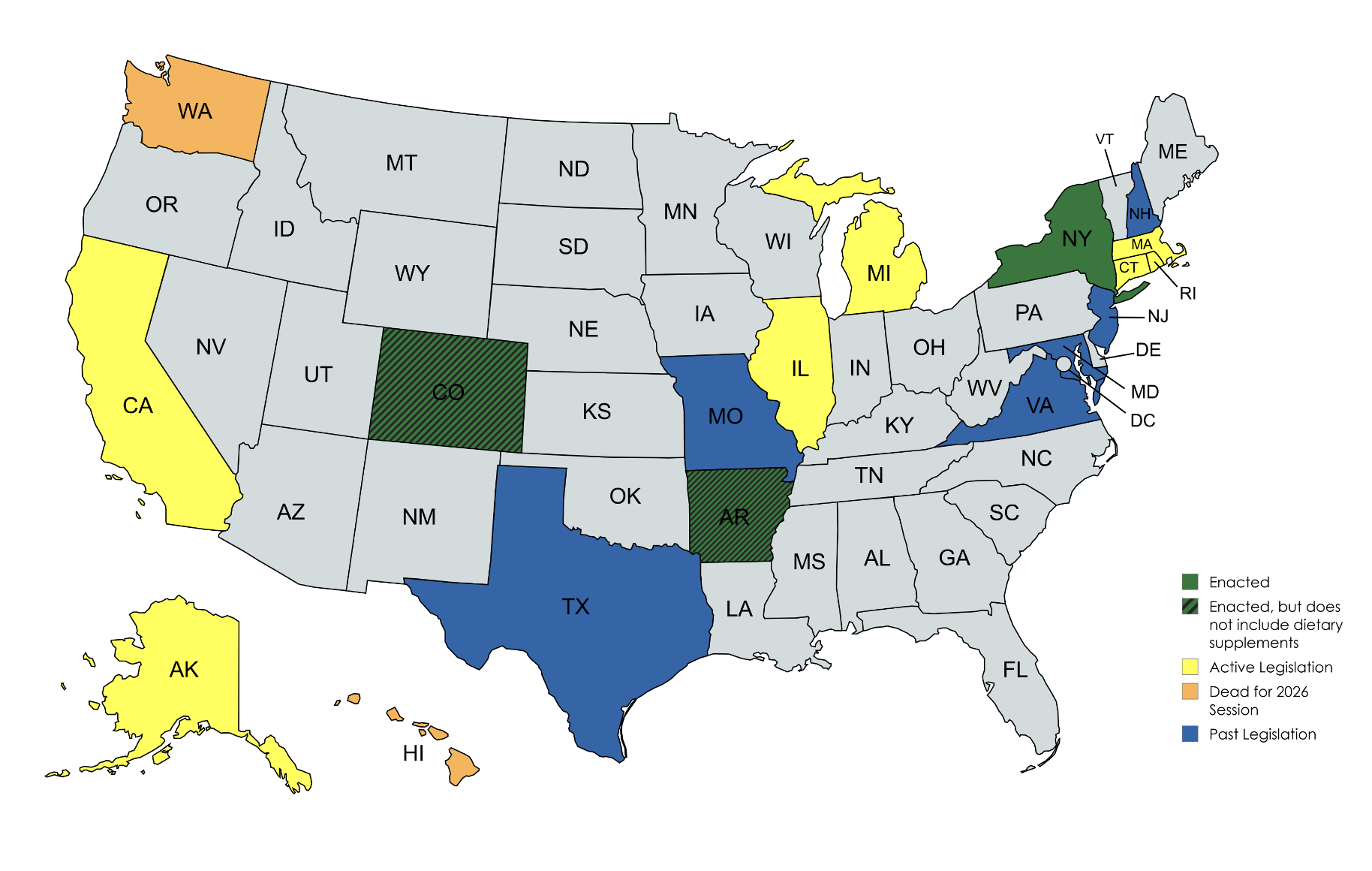 CRN is petitioning the U.S. Supreme Court to review New York’s supplement age restriction law, as similar legislation emerges in multiple states.
CRN is petitioning the U.S. Supreme Court to review New York’s supplement age restriction law, as similar legislation emerges in multiple states.
The case is worth watching closely because New York is no longer an isolated example. Similar age-restriction proposals continue to surface in multiple states, such as Rhode Island (see unlocked CRN Supplement newsletter item below), Hawaii (in which CRN testified and is now dead for 2026) which means compliance decisions made in one jurisdiction can quickly become relevant elsewhere.
CRN's litigation impacts the marketplace beyond the state of New York. It is part of a broader battle for commercial free speech with the potential to impact whether merchandising and marketing language can trigger sales bans, ID-check obligations, or locked-case requirements for otherwise lawful supplement products.
Another example of misguided state activity is California's latest protein powder bill. CRN expressed strong opposition to California Senate Bill 1033, legislation that would impose new state-specific testing, disclosure, and labeling mandates on protein powder and other concentrated protein products sold in the state.
Legislation such as SB 1033—as well as GRAS legislation cropping up in states like New York (see unlocked CRN Supplement newsletter item below) underscores why CRN strongly supports the federal Dietary Supplement Regulatory Uniformity Act, which reaffirms FDA’s authority as the national regulator of dietary supplements and prevents states from imposing conflicting or duplicative requirements. A single, science-based national standard provides clarity for consumers, consistency for manufacturers, and a stable environment for innovation and compliance.
Not plugged into updates like these?
Join the CRN member community.
Questions? Contact Ed Wyszumiala (ewyszumiala@crnusa.org)

UNLOCKED for the public from the CRN SUPPLEMENT Member Newsletter edition:
On the ground in Rhode Island: CRN opposes proposal seeking age restrictions on certain dietary supplements

In Rhode Island, S.2774 was heard on April 14 before the Senate Committee on Commerce.
- The background: This bill would restrict the sale of certain weight-loss and muscle-building dietary supplements to individuals under 18 and require age verification upon delivery.
CRN submitted a written statement for the record, and CRN Senior Director of Government Relations Mike Meirovitz delivered in-person testimony in opposition to the bill, highlighting concerns about its scope and potential unintended consequences for responsible supplement access.
CRN will continue to actively engage in advocacy efforts across multiple states where similar age-restriction proposals have been introduced, working to ensure policymakers have a clear understanding of the supplement marketplace and the potential impacts of such measures.
NY GRAS bills raise concerns; members urged to engage
There have been updates to two New York State legislative proposals—A.1556 and S.1239—that would significantly alter the regulatory framework for food ingredients, including substances determined to be generally recognized as safe (GRAS).
What's happening:
- S.1239 was advanced on March 23, when it was repassed in the Senate following an amendment and then referred to the Assembly Ways and Means Committee.
- A.1556 is currently being considered by the full Assembly; it is possible that both bills could see movement in the near term.
Why it matters: The legislation establishes a new state-level framework for GRAS substances. Specifically, it would make it unlawful to sell or use a GRAS substance in food in New York unless a detailed report has been submitted to the state and made publicly available in a state-managed database. This report would be required to include extensive information, such as safety data, intended conditions of use, dietary exposure estimates, and a comprehensive scientific basis supporting the GRAS determination. The bill further requires that such information be based on publicly available data and authorizes the state to make these reports accessible to consumers through an online database.
Yes, and: As drafted, these bills would prohibit the manufacture, sale, and distribution of food products in New York that contain certain specified additives beginning one year after enactment. The bill would also prohibit the sale of foods containing a broader list of synthetic color additives in public schools across the state.
What this means: Taken together, these provisions would create a state-specific regulatory regime for ingredient safety and GRAS determinations, separate from the existing federal framework.
CRN advocacy: CRN has taken several actions in response. We have submitted an opposition letter to the New York State Senate outlining our concerns with the legislation. In addition, CRN joined a coalition letter emphasizing the importance of maintaining a science-based, nationally uniform approach to ingredient safety. The coalition letter raises concerns that these proposals would create a patchwork of state requirements and effectively supplant the U.S. Food and Drug Administration’s authority over ingredient safety determinations.
Take action: If your company operates in or has business interests in New York, we encourage you to share your perspective with members of the New York State Legislature. As part of a broader effort, you may choose to use this advocacy tool from Americans for Food and Beverage Choice to convey opposition.
Get need-to-know updates like these in YOUR inbox every week. Join the CRN member community.
Questions? Contact Ed Wyszumiala (ewyszumiala@crnusa.org)


